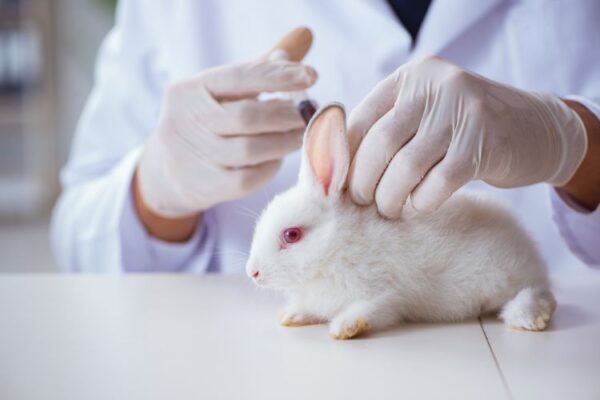
European Pharmacopoeia Eliminates Rabbit Pyrogen Test, Embraces Animal-Free Endotoxin Testing from 2026
The European Pharmacopoeia (Ph. Eur.) has announced a landmark regulatory transition in pyrogen testing, marking a decisive move away from animal-based methods. Effective 1 January 2026, the Rabbit Pyrogen Test (RPT) under general chapter 2.6.8 Pyrogens will be completely removed from all Ph. Eur. texts. This regulatory change reflects a broader European commitment to scientific…