New Approach Methodologies (NAMs), including organoids, organs-on-a-chip, microphysiological systems, and advanced computational modeling, are increasingly gaining attention in toxicology. Designed to reduce reliance on animal testing and improve the human relevance of laboratory studies, NAMs promise a faster, cost-effective way to study chemical toxicity.
However, recent analyses by Hartmut Jaeschke and Anup Ramachandran in Toxicological Sciences (Volume 208, Issue 1, 2025) highlight the limitations and challenges of fully replacing in vivo experiments with NAMs.
What Are NAMs?
NAMs encompass advanced in vitro and in silico methods. Unlike traditional 2D cell cultures, these systems aim to replicate human tissue architecture, organ-level interactions, and cellular complexity. Organoids and organs-on-a-chip allow for dose- and time-dependent toxicity testing of multiple chemicals and mixtures, offering insights into mechanisms of toxicity at unprecedented speeds. Computational modeling further accelerates toxicity predictions by simulating chemical interactions and responses.
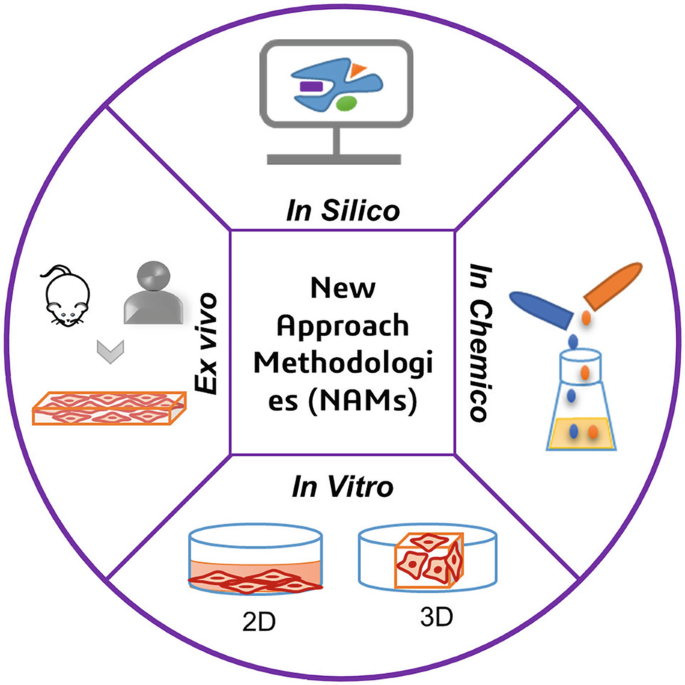
The Promise and the Pitfalls
The primary advantage of NAMs is their use of human cells, which makes results potentially more relevant to humans than animal models. They also allow multiple experimental conditions to be tested simultaneously, saving time, resources, and animals.
Yet, challenges remain. Obtaining sufficient primary human cells is difficult, and alternatives such as immortalized cell lines or induced pluripotent stem cells (iPSCs) have gene expression patterns and metabolic capacities that differ from actual human tissues. Key factors such as oxygen gradients, cellular interactions, inflammatory responses, and metabolic competency are difficult to replicate in vitro, raising questions about the direct translation of NAM results to human pathophysiology.
Why Animal Studies Still Matter
Contrary to the belief that animal testing is outdated, in vivo experiments remain essential. They provide critical insights into systemic responses, inter-organ interactions, and complex mechanisms of drug toxicity. For instance, studies on acetaminophen (APAP) overdose have shown that mouse models closely replicate human liver injury mechanisms, helping in the development of life-saving antidotes like N-acetylcysteine and fomepizole.
Bridging the Gap Between NAMs and Human Pathophysiology
Experts emphasize that NAMs should complement, not replace, animal studies. A multi-tiered approach combining NAMs with well-designed animal experiments ensures more reliable predictions of human responses. Refining NAMs based on insights from animal studies, especially using human-relevant models, is critical to advancing toxicity testing and drug discovery.
The Way Forward
While NAMs hold tremendous potential to reduce animal use and accelerate mechanistic research, their current limitations highlight the need for caution. The future of toxicology lies in integrating NAMs with high-quality in vivo research, improving reproducibility, and maintaining human relevance.
By coordinating advanced NAM technologies with carefully designed animal experiments, scientists can improve drug safety assessment, reduce unnecessary animal testing, and move closer to fully human-relevant toxicology models.
Keywords: New Approach Methodologies, NAMs, toxicology, drug hepatotoxicity, organoids, organs-on-a-chip, in vitro toxicity, in vivo experiments, acetaminophen toxicity, human-relevant models


