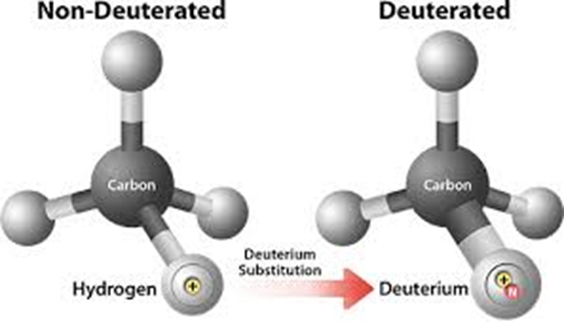
The pharmaceutical landscape has witnessed growing interest in deuterated drugs, a class of therapeutics developed by replacing specific hydrogen atoms in drug molecules with deuterium, a naturally occurring, stable, and non-radioactive isotope of hydrogen. While chemically similar to hydrogen, deuterium possesses an additional neutron, resulting in stronger carbon–deuterium bonds and measurable effects on drug metabolism and pharmacokinetics .
Scientific Background and Rationale
Deuterium was first identified in 1932, a discovery that later earned the Nobel Prize in Chemistry. Its application in pharmaceutical sciences stems from its ability to modulate metabolic pathways without altering the core chemical structure or pharmacological target of a drug. This strategic substitution has been shown to slow metabolic breakdown, particularly reactions mediated by hepatic cytochrome P450 enzymes .
The first regulatory milestone in this area occurred in 2017 with the US FDA approval of deutetrabenazine, a deuterated version of tetrabenazine, for the treatment of movement disorders associated with Huntington’s disease. This approval significantly accelerated interest in deuterium-based drug optimization .
Impact of Deuteration on Drug Metabolism
A defining feature of deuterated drugs is the kinetic isotope effect, whereby stronger carbon–deuterium bonds resist enzymatic cleavage. This can result in:
- Reduced metabolic clearance
- Increased bioavailability
- Prolonged systemic exposure
- Lower formation of potentially toxic metabolites
Such properties may enable less frequent dosing, improved patient compliance, and greater consistency in plasma drug concentrations, an important consideration for drugs with narrow therapeutic windows .
In some cases, deuteration may also subtly influence receptor binding or target selectivity, potentially enhancing therapeutic precision without changing biological activity .
Pharmacological and Safety Implications
The increased metabolic stability of deuterated compounds can translate into more predictable pharmacokinetic profiles. By minimizing fluctuations in drug levels, these compounds may reduce peak-related adverse effects while maintaining therapeutic efficacy .
From a toxicological perspective, several studies suggest that deuterated drugs may exhibit lower systemic toxicity than their non-deuterated counterparts. This is attributed to decreased formation of harmful metabolites rather than an intrinsic reduction in molecular toxicity .
Notably, experimental evidence indicates that deuterium itself demonstrates low biological toxicity at moderate exposure levels. Animal studies and limited human data have shown tolerance to partial deuterium replacement in body fluids, with adverse effects observed only at substantially high and chronic exposure levels .
Role of Deuterium in Bioanalytical Sciences
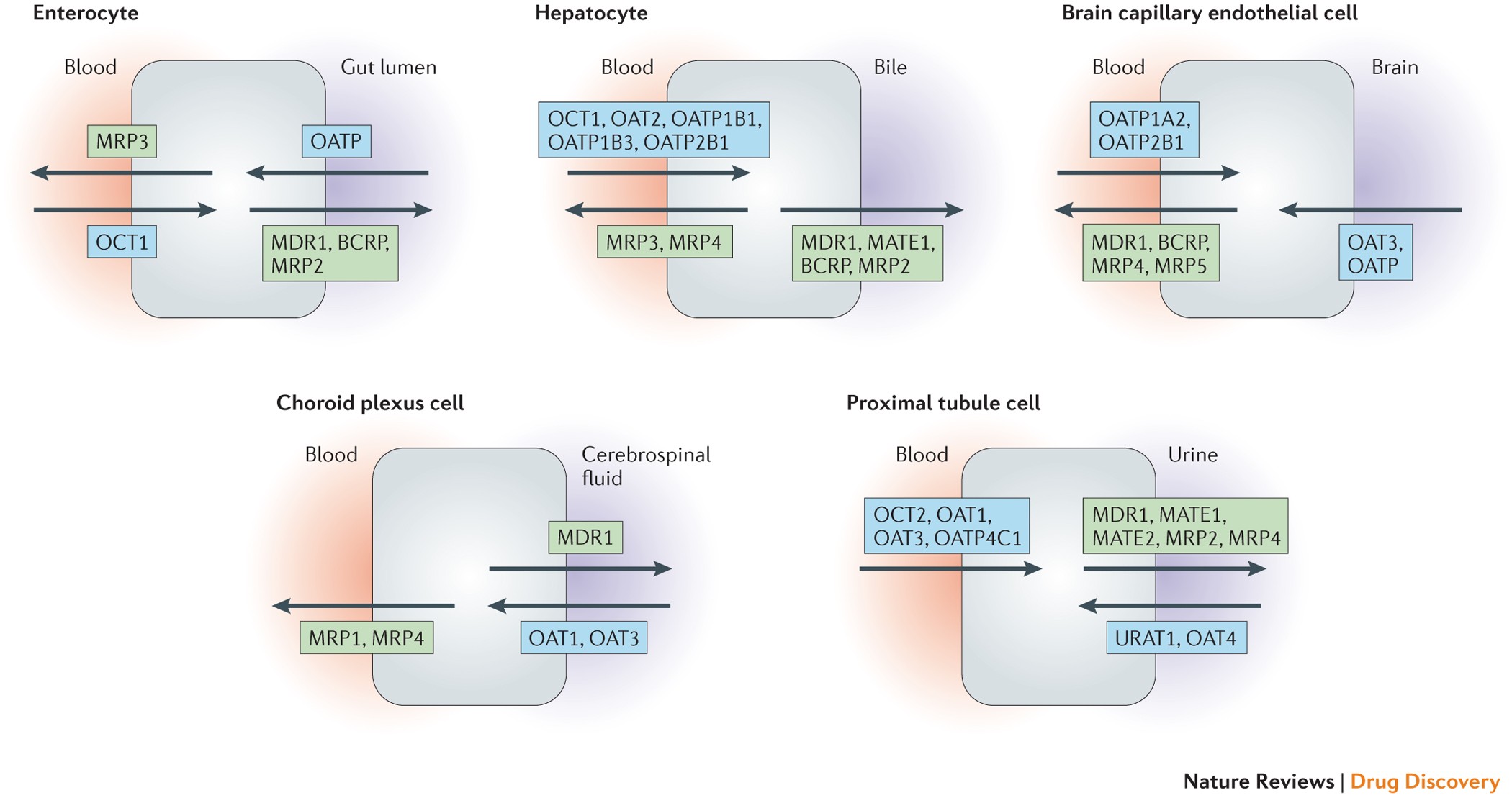
Beyond therapeutics, deuterium plays a critical role in bioanalysis, particularly as an internal standard in LC-MS/MS assays. Deuterated standards co-elute with analytes, allowing accurate correction for matrix effects, ion suppression, and extraction variability. This application has become a cornerstone in ADME studies, metabolomics, and clinical pharmacokinetic evaluations .
Safety Assessment Strategies: Multiple Development Scenarios
The safety evaluation of deuterated drugs remains context-dependent. Regulatory expectations may vary based on development strategy:
- Deuterated analogues of approved drugs are generally regarded as metabolically distinct entities, often requiring a full nonclinical toxicology package.
- For new chemical entities (NCEs) incorporating deuterium, comprehensive IND/NDA-enabling studies, including reproductive and carcinogenicity assessments, are typically expected.
- In selected oncology settings, regulators may consider bridging approaches, provided human exposure to the deuterated compound does not exceed previously evaluated levels for the undeuterated drug. Such cases often involve early regulatory engagement, including pre-IND meetings .
Regulatory Position and Market Exclusivity
The US FDA generally considers deuterated drugs as distinct active moieties, as deuteration alters covalent bond properties. Consequently, these products may qualify for five-year New Chemical Entity exclusivity, offering strategic commercial advantages alongside potential development efficiencies .
Outlook
Deuterated drugs occupy a unique intersection between medicinal chemistry, toxicology, and regulatory science. While they offer promising opportunities to improve existing therapies, their development demands careful safety assessment and transparent regulatory dialogue. As more deuterated molecules enter clinical pipelines, consistent scientific and regulatory frameworks will be essential to fully realize their potential.