Introduction
In modern toxicology and pharmaceutical science, understanding how chemicals and drugs interact with the human body goes far beyond metabolism alone. A less visible, but equally critical, ayer of biological control lies in membrane transporters. These specialized proteins act as molecular gatekeepers, regulating the movement of endogenous substances, xenobiotics, and pharmaceuticals across biological membranes. Their influence spans drug efficacy, toxicity, interindividual variability, and regulatory decision-making.
As drug development pipelines grow more complex and safety expectations become stricter, transporters have emerged as a central focus in both toxicological risk assessment and drug development strategies.
What Are Transporters and Why Do They Matter?
Transporters are membrane-bound proteins that facilitate or restrict the movement of compounds into and out of cells. Unlike passive diffusion, transporter-mediated movement is selective, regulated, and often energy-dependent.
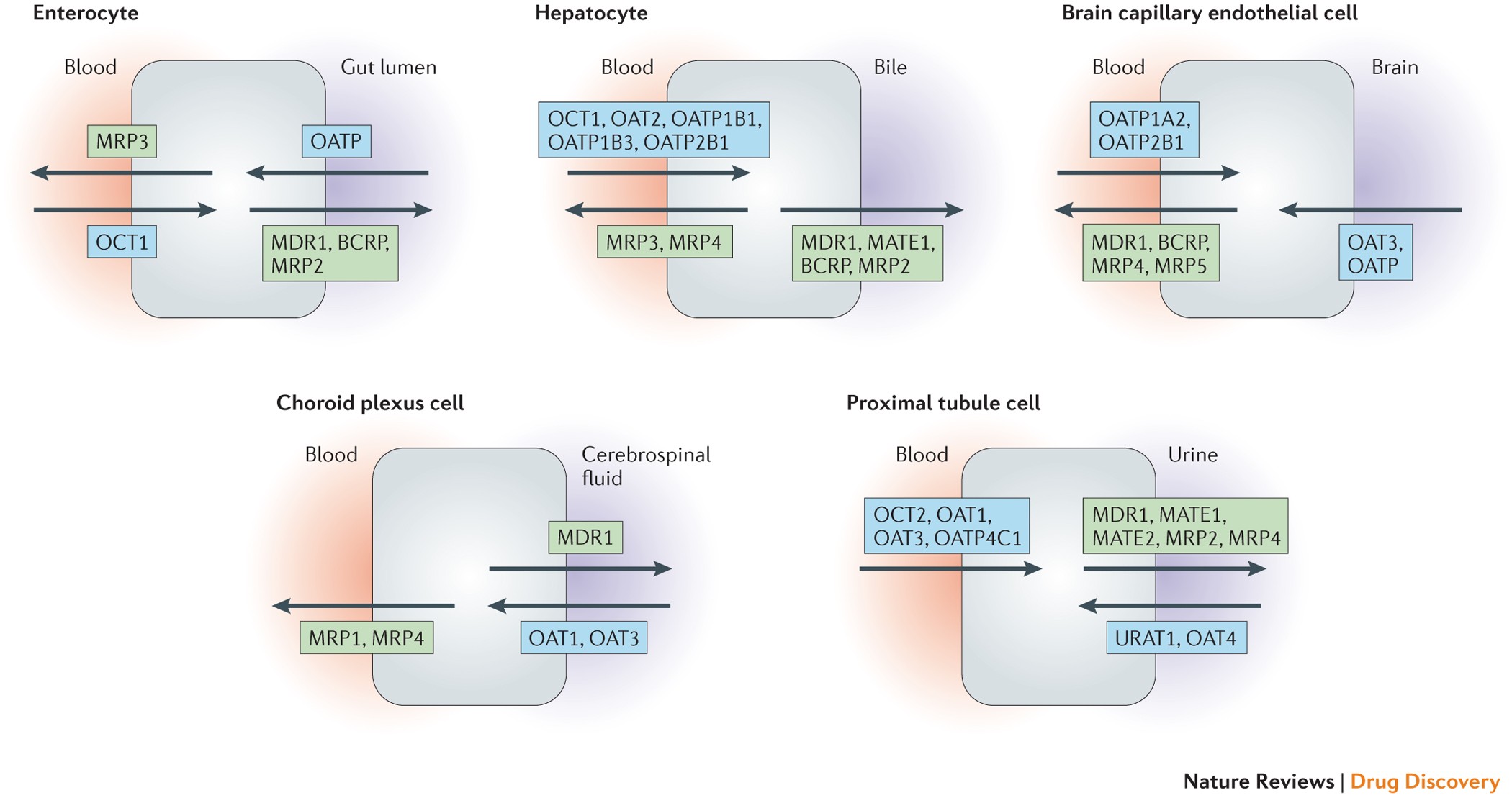
They are broadly classified into uptake transporters, which allow compounds to enter cells, and efflux transporters, which actively pump substances out, often as a protective mechanism. These transport systems are highly expressed in critical barrier tissues such as the liver, kidney, intestine, brain, and placenta. Due to this strategic localization, transporters strongly influence absorption, distribution, accumulation, and elimination of toxicants and drugs.
Transporters as Determinants of Toxicity
From a toxicological perspective, transporters can either amplify or reduce toxicity. Enhanced uptake of harmful substances into sensitive organs can increase organ-specific toxicity, while efflux transporters may protect tissues by limiting intracellular accumulation. In situations where transporters become saturated or inhibited, internal exposure may rise unexpectedly, leading to adverse effects.
This mechanism is particularly relevant for environmental contaminants, pesticides, industrial chemicals, and heavy metals. In many cases, toxicity is not driven solely by the inherent hazard of a substance, but by how efficiently transporters deliver it to vulnerable cells.
Role in Drug Development and Safety Evaluation
In drug development, transporters play a decisive role in determining oral bioavailability, tissue distribution, clearance, and overall exposure. Ignoring transporter interactions during early development has contributed to late-stage clinical failures, unexpected safety signals, and regulatory setbacks.
Today, transporter assessments are increasingly integrated into lead optimization, nonclinical safety studies, and clinical pharmacokinetic evaluations. Early identification of transporter involvement improves human exposure predictions and supports the development of safer and more effective therapeutic products.
Transporter-Mediated Drug–Drug Interactions
Transporter-related drug–drug interactions represent a significant safety concern. When multiple drugs compete for or inhibit the same transporter, systemic exposure to one or more compounds may increase, even at standard dosing levels. This can elevate the risk of toxicity, particularly for drugs with narrow therapeutic margins.
Such interactions are now a key area of regulatory focus, emphasizing the need for systematic transporter evaluation during drug development and post-marketing surveillance.
Genetic Variability and Individual Susceptibility
Transporter expression and function can vary considerably among individuals due to genetic polymorphisms, disease conditions, age, and physiological status. This variability helps explain why certain individuals experience adverse effects at normal doses while others exhibit reduced therapeutic responses.
In toxicology, these differences underscore the importance of considering susceptible populations and support the transition toward personalized and population-specific risk assessment approaches.
Regulatory Perspective and Emerging Expectations
Regulatory authorities worldwide increasingly expect transporter-related data as part of drug approval dossiers, safety pharmacology packages, and chemical risk assessments. Transporter evaluation is no longer viewed as supplementary information but as an essential element of modern safety science, influencing labeling decisions, clinical study design, and risk management strategies.
Future Outlook
Advances in in vitro transporter models, in silico prediction tools, and systems toxicology approaches are improving the integration of transporter data into regulatory and scientific decision-making. Combining transporter biology with exposure science, genomics, and computational modeling offers a more predictive and human-relevant framework for evaluating chemical and drug safety.
Conclusion
Transporters represent a critical yet often underrecognized component of toxicology and drug development. By shaping internal exposure rather than external dose alone, they redefine how safety and efficacy are understood. As toxicology continues to evolve toward mechanism-based and human-relevant methodologies, transporters will remain central to improving drug development outcomes and protecting public health.