Sixth Edition of ISO 10993-1 Released With Stronger Focus on Chemical Risk Mitigation
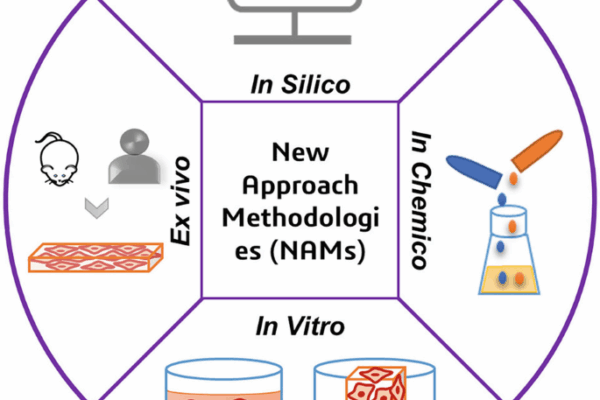
The much-anticipated sixth edition of ISO 10993-1, the global standard guiding the biological evaluation of medical devices, has officially been released. The updated standard aligns more closely with the risk-based approach of ISO 14971, introducing strengthened emphasis on identifying and mitigating chemical risks associated with medical devices. The revised ISO 10993-1 outlines enhanced principles for…